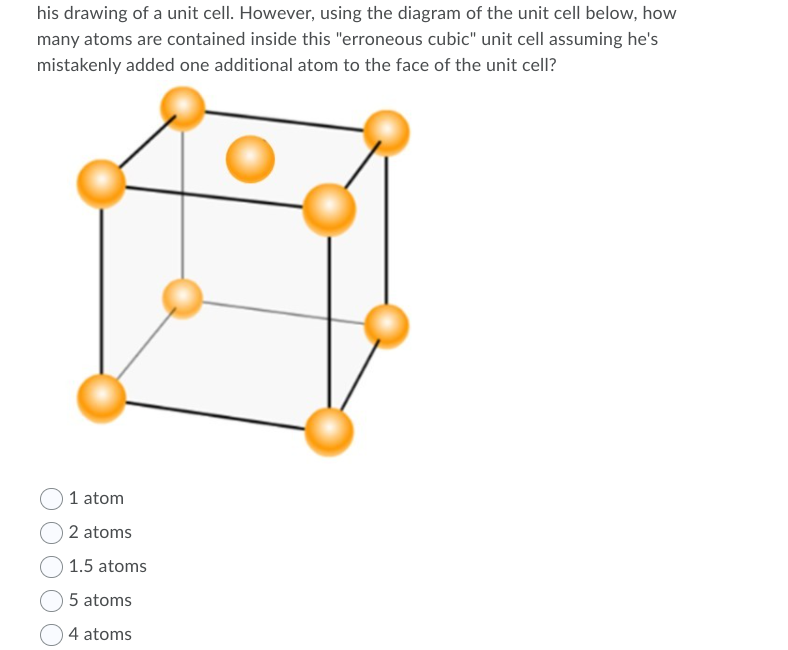
Formula of unit cell = PQ 2. Calculate number of atoms in a cubic unit cell having one atom on each corner and two atom on each body diagonal. There are total

Atomic structure of various carbides. (a) Unit cell of one ω′-Fe 6 C... | Download Scientific Diagram











/GettyImages-909208550-ca787b9519ca4cf6bc6ade14a0243fb8.jpg)

:max_bytes(150000):strip_icc()/GettyImages-85976214-56a1341d3df78cf772685c8e-5c2d3cf3c9e77c0001944896.jpg)